Overview:
Rabeprazole is a medication used to treat acid reflux, heartburn, and symptoms of gastroesophageal reflux disease, among others. It is classified as a proton pump inhibitor (PPI) and acts by lowering the quantity of acid produced by the stomach. Rabeprazole is available as a delayed-release oral capsule and enteric-coated oral tablets. The US Food and Drug Administration (FDA) approved Rabeprazole for acid reflux treatment in 1999.
How Does Rabeprazole Work?
Rabeprazole is a prodrug proton pump inhibitor that gets converted into active sulphenamide form in the acidic environment of the stomach's parietal cells. It works by blocking the action of H+ and K+ ATPase, and the medication suppresses both basal and induced gastric acid output in a dose-dependent manner. Hence, it reduces the amount of acid produced in the stomach, which helps in relieving the pain from acid-related indigestion and heartburn.
Dosage:
-
The starting dose of Rabeprazole is 10 mg.
-
Administer the drug as instructed by the physician.
-
For Delayed-Release Tablets: Do not chew, split, or crush the tablet. This medication can be used with or without food. Follow the doctor's instructions if they specify how to take the medication.
-
For Children Taking Delayed-Release Capsules:
-
Do not swallow the delayed-release capsule as a whole.
-
Take the medication at least twenty to thirty minutes before the meal.
-
If using a liquid, such as infant formula, apple juice, or pediatric electrolyte solution, open the capsule and pour the contents into a small quantity of liquid or soft food (such as juices, applesauce, fruit, yogurt, or vegetable baby food).
-
Take the medication within 15 minutes. Do not chew the mixture; swallow it. Do not hold it for future use.
Warnings:
-
Acute Interstitial Nephritis:
Patients on PPIs, particularly Rabeprazole, seem to develop acute interstitial nephritis. During PPI medication, acute interstitial nephritis can happen at any time and is typically attributable to an idiopathic hypersensitivity reaction. If acute interstitial nephritis appears, stop using Rabeprazole.
-
C.difficile Associated Diarrhea:
Published observational studies imply that PPI medication, such as Rabeprazole, may be linked to a higher incidence of diarrhea caused by Clostridium difficile, particularly in hospitalized patients. When diarrhea does not get better, the diagnosis of this condition is to be taken into account.
-
Increased Risk of Fractures:
PPI medication may be linked to a higher risk of hip, wrist, or spine fractures caused by osteoporosis, according to a number of published observational studies in adults. Patients who got high-dose PPI medication, which is defined as several daily doses, over an extended period of time had a higher risk of fracture (a year or longer). Patients should receive the lowest amount of PPI medication and the shortest amount of time necessary for the problem to be treated.
-
Vitamin-B12 Deficiency:
Long-term (more than three years) daily use of any acid-suppressing drugs may result in hypochlorhydria (reduction in stomach acid) or achlorhydria (absence of stomach acid), which will cause malabsorption of cyanocobalamin (vitamin B-12).
-
Low Magnesium Levels:
Patients taking proton pump inhibitors (PPIs) for at least three months have a rare chance of developing low magnesium levels, both symptomatic and asymptomatic, usually after a year of treatment. Most patients needed magnesium supplementation and PPI discontinuance as part of their treatment for hypomagnesemia.
For Patients
What Is Acid Reflux?
Acid reflux is when bile or stomach acid irritates the food pipe lining. This chronic disease occurs when stomach acid or bile comes in contact with the food pipe and irritates the lining. When acid reflux and heartburn occur, more than twice a week may indicate GERD.
The symptoms include burning or throbbing pain in the chest area, usually experienced after eating, and worsening when lying down. Other typical risk factors for acid reflux disease include the following:
-
Huge meals or, immediately following a meal, lying down.
-
Being obese or overweight.
-
Consuming a substantial meal, lying on the back, or hunching the waist.
-
Eating just before going to bed.
-
Eating specific foods, such as fatty or spicy foods, citrus, chocolate, tomato, mint, garlic, or onions.
-
Drinking specific liquids, like alcohol, fizzy beverages, coffee, or tea.
-
Smoking.
-
Being pregnant.
-
Using some muscle relaxants, Aspirin, Ibuprofen, or blood pressure drugs.
Relief from lifestyle changes and over-the-counter medication is usually temporary.
Learn More About Rabeprazole:
Before Starting Rabeprazole:
When and Why to Take Rabeprazole?
Rabeprazole is a prescription drug. Administer the medication as per the doctor's order. In case of doubts or queries concerning drug use, contact the doctor or pharmacist immediately. Speak with the doctor or pharmacist if an experience with the drug has a stronger or weaker effect than being informed.
What Are the Things to Inform The Doctor Before Taking Prescribed Rabeprazole?
Inform the doctor if any of the following conditions are present:
-
If the patient has an allergy to "substituted Benzimidazoles" or other proton pump inhibitor medications (such as Esomeprazole, Omeprazole, Lansoprazole, or Pantoprazole).
-
Have a stomach or food pipe tumor. The medical professional may order certain tests to rule.
-
Have liver disease.
-
If the patient uses antiviral medicine like Atazanavir, a medication used to treat HIV infection.
-
If the patient is on long-term Rabeprazole treatment and takes medications like Digoxin (for heart failure) or water pills such as Spironolactone, Furosemide, Hydrochlorothiazides (used to treat high blood pressure or cardiac issues).
-
If the patient takes Rabeprazole for a prolonged period and has low body reserves or risk factors for low vitamin B12 levels. The absorption of vitamin B12 may be reduced in the presence of Rabeprazole, as with all acid-reducing medications.
-
If the patient has ever experienced a skin reaction following treatment with a drug similar to Rabeprazole.
-
If the patient is scheduled for Chromogranin A blood test.
Starting Rabeprazole:
How Is Rabeprazole Given?
Things to Do After Starting Rabeprazole:
-
Some patients have experienced blood issues (such as a decrease in blood cells that could lead to fever, more frequent infections, or easy bruising and bleeding) and liver issues (such as an increase or decrease in liver enzymes that could be seen in blood tests). Rabeprazole is discontinued until these symptoms resolve.
-
Rabeprazole should be stopped immediately if the patient experiences severe (watery or bloody) diarrhea and other symptoms like fever, abdominal pain, or soreness.
-
Inform the physician immediately if the patient develops a rash on the skin, especially in sun-exposed regions, since the drug may be discontinued in such cases.
-
Long-term use (one year or more) of Rabeprazole can increase the risk of hip, wrist, or spine fracture. Hence, it is important to let the doctor know if the patient is also taking any drug belonging to the corticosteroid class (which can increase the risk of osteoporosis).
What Are the Side Effects of Rabeprazole?
It is required to report to the doctor if any side effects are experienced after taking the drug. Rabeprazole's list of side effects is as follows:
Common Side Effects:
-
Infections.
-
Trouble sleeping.
-
Headache or lightheadedness.
-
Cough, pharyngitis (a painful throat), and runny nose.
-
Effects on the stomach or digestive system include gastrointestinal discomfort, diarrhea, flatulence, nausea, vomiting, or constipation.
-
Back or joint discomfort.
-
Dry mouth.
-
Flu-like symptoms and weakness.
-
Benign stomach polyps.
-
Drowsiness.
-
Infected chest (bronchitis).
-
Blocked and uncomfortable sinuses (sinusitis).
-
Bloating.
-
Redness or a skin rash.
-
Leg cramps, muscle discomfort, or joint pain.
-
Infected bladder (urinary tract infection).
-
Chest pain.
-
Cold and fever.
Serious Side Effects:
-
Hip, wrist, or spine fracture.
-
Allergic responses might involve abrupt facial swelling, respiratory problems, or low blood pressure, which can result in fainting or collapsing.
-
Frequent infections, such as sore throats, fevers, mouth or throat ulcers, or high body temperatures. This might be caused by a decline in a particular type of white blood cell.
-
Liver issues can cause inflammation of the liver (hepatitis), which is characterized by symptoms like yellowing of the skin, and white of the eyes (jaundice), and in people with a history of liver issues, hepatic encephalopathy (brain damage due to liver disease).
Make sure to report all the serious side effects of the medication to the doctor immediately for the necessary intervention.
Dietary Alterations:
The patient may eat and normally drink while taking Rabeprazole. However, it is recommended to take it before a meal. However, avoiding fatty, spicy, and rich foods that exacerbate symptoms is advised. Additionally, the patient should consume less alcohol and caffeinated beverages like tea, coffee, and soda.
What Should Be Done if a Dose Is Missed?
Take the missed dose as soon as it is remembered. However, if the time for the next dose is almost near, skip the missed dose and go on as normal. But, if it is over five days since the last time of medication, consult the doctor before taking any additional medication. Never take two doses at once to make up for missed ones.
What Should Be Done to Treat Rabeprazole Overdose?
If overdosing on the medication happens, immediately report to the doctor or contact the poison control center at the earliest. Always carry the medication kit along so that the healthcare team will be able to counter the effects of overdosing better.
How to Store Rabeprazole?
-
Store Rabeprazole at room temperature between 15 to 30 degrees Celsius.
-
Keep the tablets away from children and pets.
-
The medication bottle must be consumed within 60 days of the initial opening. Therefore, even if there are still some tablets in the bottle after 60 days of opening, discard them. In the area provided on the bottle label, note the day of first opening it to help remember it.
-
Protect the tablets against moisture by storing them in the original container.
-
If the patient observes a change in the tablet's color while taking the medication, stop using it.
-
Avoid disposing of medications in wastewater or household garbage. Talk to the pharmacist about how to dispose of expired medications. These measures will aid in environmental protection.
Avoid Self-Medication:
Avoid taking Rabeprazole without a prescription, and do not recommend it to others. Avoid taking the medication on someone else's advice. Different people react to medications differently, and some may even react worse. Ensure to make a list of all the prescriptions and non-prescription that are being taken whenever consulting a doctor, and take the medication only as instructed by a qualified healthcare professional.
For Doctors
Indication:
The indications of Rabeprazole include
-
Gastric hypersecretion.
-
Gastroesophageal reflux disease.
-
H.pylori infection.
-
Ulcer of the duodenum.
Dosing:
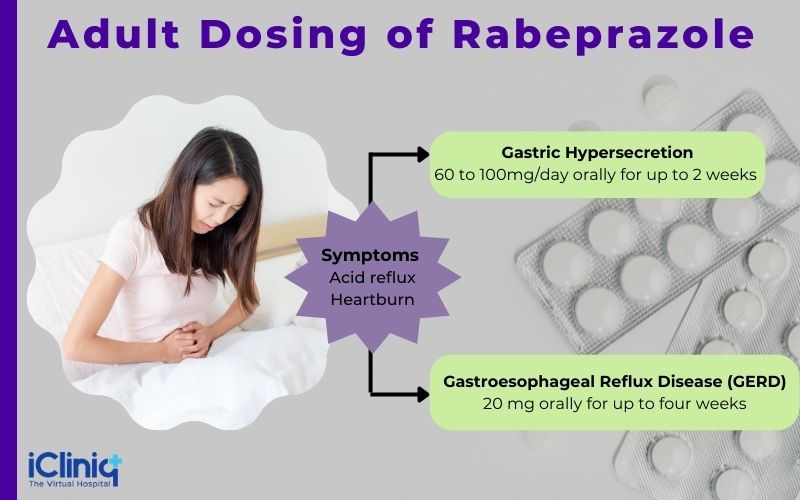
Dosing Considerations:
-
Renal and Hepatic Impairment: No dosing adjustment is required.
-
Acute Tubulointerstitial Nephritis: Discontinue use.
Pharmacology:
Mechanism of Action:
Rabeprazole sodium is a gastric proton pump inhibitor that does not possess anticholinergic or histamine H(2)-receptor antagonist properties. It suppresses gastric acid secretion by inhibiting the gastric H+ and K+-ATPase at the secretory surface of parietal cells.
Pharmacokinetics (Delayed-Release Tablets):
Absorption:
A 20 mg oral pill of Rabeprazole has an approximate 52 % absolute bioavailability (in comparison to intravenous delivery). Tmax varies when Rabeprazole delayed-release tablets are given along with a high-fat meal, and concurrent food consumption can cause the absorption to be delayed for up to four hours or more. However, there are no noticeable changes to the Cmax or the AUC of Rabeprazole. Therefore, the timing of meals is irrelevant when using Rabeprazole delayed-release pills.
Distribution:
Rabeprazole has been shown to be 96.3 % bound to human plasma proteins.
Metabolism:
The metabolization of Rabeprazole is substantial. Systemic nonenzymatic reduction converts a considerable portion of Rabeprazole into a thioether molecule. Additionally, Rabeprazole is converted by the liver's cytochrome P450 enzyme to the chemicals sulphone and desmethyl.
The main metabolites tested in human plasma are thioether and sulphone. There was no discernible antisecretory activity for these metabolites.
Excretion:
Following a single oral administration of 20 mg of 14C-labeled Rabeprazole, around 90 % of the medication was excreted in the urine, mostly as the metabolites thioether carboxylic acid, glucuronide, and mercapturic acid. The remaining dosage was found in the feces. 99.8 % of the radiation was completely recovered. There was no unaltered Rabeprazole found in the urine or feces.
Active Ingredient: The active ingredient in Rabeprazole is Rabeprazole Sodium.
Inactive Ingredient: The other ingredients are
-
Core: Mannitol, hydroxypropyl cellulose, magnesium oxide, low substituted hydroxypropyl cellulose, ethylcellulose, magnesium stearate, iron oxide red, acetylated mono and diglycerides, iron oxide yellow, hypromellose phthalate, talc, and titanium dioxide.
-
Film-Coating: Hypromellose 6cP, titanium dioxide, acetylated monoglycerides, and iron oxide red.
Toxicity:
Clinical Toxicology:
Overdose effects are anticipated to be an extension of adverse effects observed following therapeutic doses. In limited overdose cases, clinical effects have consisted of mild tachycardia, flushing, somnolence, confusion, headache, blurred vision, abdominal pain, nausea, vomiting, dry mouth, and moderate leukocytosis.
Management:
-
Management of Mild to Moderate Toxicity: Treatment is symptomatic and supportive. Correct any significant fluid or electrolyte abnormalities in patients with severe diarrhea and vomiting.
-
Management of Severe Toxicity: Treatment is symptomatic and supportive. Significant toxicity is not expected after an overdose.
Warnings and Precautions:
Beers Criteria:
-
Unless necessary for high-risk patients (for example, oral corticosteroids, chronic NSAID use), erosive esophagitis, pathological hypersecretory condition, Barrett's esophagus, or need for maintenance treatment. For example, due to failure of drug discontinuation or histamine-2 blockers, avoid scheduled use in older adults for longer than eight weeks.
Dermatologic:
-
Proton pump inhibitors have been linked to severe cutaneous side effects, such as Stevens-Johnson syndrome, toxic epidermal necrolysis, and acute widespread exanthematous pustulosis; discontinuation is necessary in such cases.
-
Within weeks to years after continued medication therapy, cases of new or worsening cutaneous lupus erythematosus have been reported; avoid using for longer than medically necessary and stop using if suspected.
Endocrine and Metabolic:
-
With prolonged use (for example, more than one to two years), vitamin B12 insufficiency may result; monitoring is advised.
-
In patients on proton pump inhibitors for at least three months, with most instances occurring after a year of treatment, hypomagnesemia, both symptomatic and asymptomatic, has been recorded infrequently; severe side effects include tetany, arrhythmias, and seizures.
-
Monitoring is advised with extended therapy or concurrent use of medications that produce hypomagnesemia.
-
Patients with a preexisting risk of hypocalcemia (such as hypoparathyroidism) may experience hypomagnesemia, which can cause hypocalcemia and hypokalemia and exacerbate underlying hypocalcemia; monitoring is advised, and supplementation with magnesium and calcium as well as discontinuation may be necessary.
Gastrointestinal:
-
Rabeprazole use is associated with a potential risk of fundic gland polyps that increases with long-term use, especially beyond one year; use the shortest duration of therapy appropriate to the condition being treated.
-
The symptomatic response does not preclude the presence of gastric malignancy in adults.
-
Clostridium difficile-associated diarrhea may occur, especially in hospitalized patients; use the lowest dose and shortest duration.
Immunologic:
-
Proton pump inhibitors have been linked to severe cutaneous side effects, including eosinophilia and other systemic symptoms; discontinuation is necessary in such cases.
-
Within days to years of starting treatment, cases of new or worsening systemic lupus erythematosus have been documented; avoid using longer than is medically necessary and stop using if such reactions are suspected.
Musculoskeletal:
-
Use the lowest dose and shortest treatment period possible to prevent osteoporosis-related bone fractures of the hip, wrist, or spine, especially with larger (multiple daily) doses or longer therapy durations (one year or more).
Renal:
-
Proton pump inhibitor use has been linked to acute tubulointerstitial nephritis, which can happen at any time during treatment. Discontinue the drug in such cases.
Contraindications:
Rabeprazole is contraindicated in the following conditions:
-
History of hypersensitivity to Rabeprazole, substituted benzimidazoles, or any of the product's ingredients, including anaphylaxis, anaphylactic shock, acute tubulointerstitial nephritis, angioedema, bronchospasm, and urticaria.
-
Concomitant use of Rilpivirine-containing products.
-
Concurrent use with Nilfenavir.
Clinical Studies:
In a multicenter, randomized, double-blind, placebo-controlled research conducted in the United States, 103 patients were given a placebo, delayed-release Rabeprazole tablets containing 10 mg, 20 mg, or 40 mg once daily for up to eight weeks. Only individuals with GERD (gastroesophageal reflux disease) symptoms and at least grade 2 esophagitis (according to the modified Hetzel-Dent grading scale) were allowed to participate in this study, and all others on GERD healing. Grade 0 or 1 endoscopic healing was specified. After four and eight weeks of treatment, each Rabeprazole dose demonstrated significant effects from the placebo in terms of promoting endoscopic healing.
Results:
All Rabeprazole groups reported considerably greater rates of complete betterment of the severity of GERD daytime heartburn when compared to placebo at weeks four and eight. The mean reductions from baseline in the dose of daily antacid were statistically significant for all Rabeprazole groups compared to placebo at weeks four and eight.
Drug Interactions:
The drug interactions of Rabeprazole include
-
Acalabrutinib.
-
Amphetamine.
-
Atazanavir.
-
Atezolizumab.
-
Belumosudil.
-
Benzphetamine.
-
Bosutinib.
-
Capecitabine.
-
Cilostazol.
-
Clopidogrel.
-
Dacomitinib.
-
Dasatinib.
-
Dextroamphetamine.
-
Digoxin.
-
Erlotinib.
-
Eslicarbazepine Acetate.
-
Gefitinib.
-
Infigratinib.
-
Ketoconazole.
-
Ledipasvir.
-
Lisdexamfetamine.
-
Methamphetamine.
-
Methotrexate.
-
Mycophenolate Mofetil.
-
Nelfinavir.
-
Neratinib.
-
Nilotinib.
-
Octreotide.
-
Palbociclib.
-
Pazopanib.
-
Pexidartinib.
-
Saquinavir.
-
Secretin Human.
-
Selpercatinib.
-
Sotorasib.
-
Sunitinib.
-
Velpatasvir.
-
Vismodegib.
-
Levothyroxine.
Other Specifications:
Rabeprazole in Pregnant Women:
There are no well-controlled and adequate studies of Rabeprazole in pregnant women. Rabeprazole has not been reported with any evidence of teratogenicity in animal reproduction studies at thirteen and eight times the human exposure at the recommended dose for GERD in rabbits and rats, respectively.
Rabeprazole in Lactating Women:
Although it is unknown whether Rabeprazole is excreted in human milk, it is found in rat milk. When administering Rabeprazole to a breastfeeding woman, attention should be taken because many medications are secreted in milk.
Rabeprazole in Geriatric Patients:
Although it cannot be ruled out that some older people may be more sensitive than others, there were no overall differences in safety or effectiveness between the geriatric subjects and younger subjects, and other reported evidence from clinical experience has not identified differences in responses between the elderly and younger patients.












