Overview:
Linaclotide is a drug that is used to treat the condition of chronic irritable bowel syndrome or constipation. The drug increases intestinal fluid secretion, which further helps ease the passage of stools and relieve the symptoms of constipation. Chemically Linaclotide is a heterodetic cyclic peptide that contains 14 amino acids. The drug belongs to the class of drugs called IBS (irritable bowel syndrome) agents, gastrointestinal and guanylate cyclase-C (GC-C) agonists. The FDA (United States Food and Drug Administration) approved the drug on August 30, 2012, for treating irritable bowel syndrome.
Uses of the Drug Linaclotide:
The drug treats certain types of bowel diseases such as irritable bowel syndrome and chronic idiopathic constipation. As stated, the drug increases the secretion of intestinal fluids, which helps to increase the speed of food in the intestine. Linaclotide helps to improve the symptoms like abdominal pain, bloating, straining, and the feeling of incomplete bowel movements.
Warnings:
-
The drug should not be administered if there is a history of blockage in the intestine.
-
Linaclotide is not advised in children less than 18 years of age.
-
A child younger than six should not use Linaclotide as it may lead to fatal dehydration due to severe diarrhea.
About the Disease:
What Is Irritable Bowel Syndrome?
Irritable bowel syndrome is a common disorder that may affect the large intestine. The symptoms of the disease include abdominal pain, cramping, gas, bloating, diarrhea, or constipation. The condition can be chronic and may require long-term management. However, the symptoms of irritable bowel syndrome can only be severe in a few cases. Many people manage their issues well by following a proper diet, maintaining a lifestyle, and avoiding stress. The syndrome does not affect the lining and tissues of the intestine and does not lead to colorectal cancer.
Some of the severe symptoms of irritable bowel syndrome are:
-
Weight loss.
-
Rectal bleeding.
-
Iron deficiency anemia.
-
Dysphagia (difficulty in swallowing).
-
Unexplained consistent vomiting.
More About the Drug:
For Patients:
Before Starting the Drug: The doctor should be informed about some important conditions before starting the drug. These include the presence of any drug allergy or any blockage in the intestinal region. The drug should be administered according to an individual's age and body weight and should not be given to children under 18 years of age. The drug is advised to be avoided completely in children less than six years of age as it may lead to severe diarrhea and fatal dehydration.
How Is Linaclotide Taken:
-
Before starting the drug, the instructions mentioned on the label are to be followed as it is. Linaclotide should be taken strictly on the doctor's advice. It is not advised to take the drug on your own.
-
The drug should be taken early in the morning on an empty stomach at least 30 minutes before the first meal.
-
The capsule should be swallowed whole, not chewed, crushed, or opened.
-
If there is difficulty in swallowing the capsule completely, it is advised to open the capsule and sprinkle it with a spoon of water which should be swallowed immediately and not left for later. Linaclotide taken by this technique should also be consumed 30 minutes before the meal.
-
The symptoms subside at least after two weeks of consumption of the Linaclotide. So it is important to keep taking the medicine until the doctor has advised.
Storage of the Drug:
The drug should be kept at room temperature away from moisture and heat. The tablets should be kept always in their original containers along with a canister of moisture-absorbing preservatives. The medicine should be stored out of the reach of children as it can be fatal for them if accidentally swallowed.
What Happens if a Dose of Linaclotide Is Missed?
If any dose of the drug is missed, it is advised to take the other dose at its time and not consume two doses at a time as it may lead to side effects.
Side Effects of Linaclotide: The drug can have some side effects depending on its doses and the body status of the individuals.
Some of the common side effects shown by the drug are:
-
Diarrhea: Loose stools, which are very frequent during the day.
-
Abdominal pain, which is particularly stomach pain.
-
Gas or bloating of the stomach.
-
Feeling of fullness in the stomach.
These mentioned side effects are normal for the drug and may subside eventually; however, some of the severe side effects shown by Linaclotide are:
-
Allergic reactions such as hives (urticaria), difficulty in breathing with swelling of face, lips, tongue, and throat.
-
Ongoing diarrhea which gets severe eventually leading to weakness and fatigue. The person may also suffer severe dehydration due to frequent episodes of diarrhea.
-
Feeling of dizziness and light-headedness. The person may feel like passing out at any time.
-
Irregular heartbeats and muscle weakness.
-
Acute leg cramps due to muscle weakness.
-
Stool with blood stains which can be red or black (digested blood).
These are some of the side effects of Linaclotide, however, it can show some other unpredicted symptoms too, so it is important to keep a watch on the conditions and inform the doctor immediately if there is any serious issue.
Doses of Linaclotide:
The doses of the drug depend on the condition it is treating.
-
Adult doses for Irritable bowel syndrome are 290 mcg orally once a day. The capsule should be taken 30 minutes prior to the first meal of the day without crushing or chewing. If the patient has difficulty swallowing the capsule, it is recommended to either open the capsule and mix it with water or administer nasogastrically through the gastrostomy tube.
-
In cases of irritable bowel syndrome with constipation the dose is 145 mcg orally once every day. In some patients, the dose can be 72 mcg orally once a day depending on the individual's characteristics and tolerability. The drug administration method can be similar to in cases of irritable bowel syndrome.
For the Doctor:
Indications: Linaclotide is a peptide agonist of guanylate cyclase 2C administered orally to treat irritable bowel syndrome and chronic idiopathic constipation. The drug is a chemically heterodetic cyclic peptide that contains fourteen amino acids. The protein sequence of the drug is Cys Cys Glu Tyr Cys Cys Asn Pro Ala Cys Thr Gly Cys Tyr.
Pharmacodynamics: The changes in the appearance and consistency of the stool is measured by the BSFS (Bristol stool form scale) after the consumption of linaclotide. The Bristol stool chart is a clinical assessment of the stool in seven different types. The chart helps diagnose many issues like diarrhea, constipation, or irritable bowel syndrome.
The chart is classified as follows:
-
Type 1 and type 2 indicate constipation as an issue.
-
Type 3 and type 4 are easier to pass and are considered ideal stools.
-
Type 5, 6, and 7 indicate diarrhea and urgency for treatment.
Mechanism of Action:
-
As stated Linaclotide is an agonist of guanylate cyclase-C, the drug, and its active metabolite when bound with guanylate cyclase-C, produce a local effect on the luminal surface of the intestinal epithelium.
-
This activation of guanylate cyclase-C by Linaclotide leads to an intracellular and extracellular increase in the cyclic guanosine monophosphate concentrations (cGMP).
-
The elevation of this cGMP stimulates the secretion of bicarbonate and chloride in the intestinal lumen by the activation of cystic fibrosis transmembrane conductance regulator (CFTR) ion channels.
-
So ultimately, Linaclotide helps the cases of irritable bowel syndrome as gastrointestinal transit is accelerated and the release of intestinal fluid is increased.
-
In the cases of animal models, it was noticed that there was a decrease in visceral pain after the administration of Linaclotide. This decrease in pain-sensing activity can be due to an increase in extracellular cGMP.
Absorption: Linaclotide, when taken orally, does not absorb in systemic regulation. It was observed that even after consumption of 125 mcg or 290 mcg of the drug, there were no traces of active metabolites or the drug in the systemic circulation.
Volume of Distribution: As the drug does not enter the system circulation, the plasma concentration of the drug can not be measured. The drug is expected to minimally distribute to the tissues.
Metabolism: Linaclotide is metabolized within the gastrointestinal tract to its principal, active, and metabolite states. The drug Linaclotide and its active metabolite are proteolytically degraded within the intestinal lumen into smaller peptides and naturally occurring amino acids.
Route of Elimination: Linaclotide is eliminated in the feces with 3 % to 5 % of active metabolites. Most doses of Linaclotide undergo proteolysis (reduction of disulfide bonds) in the intestines before excretion.
Half-Life of Linaclotide: As the drug is not absorbed systemically, it is difficult to calculate its half-life.
Adverse Reactions: The most common adverse reaction of Linaclotide in irritable bowel syndrome or constipation patients are abdominal pain, diarrhea, abdominal distension, and flatulence.
Drug Interactions: The interaction of Linaclotide with other drugs are stated as:
-
Amitriptyline: The therapeutic efficacy of Linaclotide is decreased when it is used in combination with Amitriptyline.
-
Atropine: It is known to decrease the efficacy of Linaclotide.
-
Benzatropine: The drug reduces the therapeutic efficacy of Linaclotide.
-
Benzthiazide: The drug may increase the risk of adverse effects due to Linaclotide when used in combination.
-
Chloroprocaine: The drug decreases the therapeutic efficacy of Linaclotide.
-
Furosemide: The chances of adverse effect increases when the drug is combined with Linaclotide.
-
Magnesium: It reduces the efficacy of Linaclotide in combination.
Clinical Trials of Linaclotide:
-
Trials of Linaclotide in patients with chronic constipation: this trial is performed to determine the efficacy and safety of Linaclotide in the treatment of cases of chronic constipation. The parameter which is used primarily is the percentage of patients from each dosing group who meet the protocol definition for a complete spontaneous bowel movement (CSBM).
Number of Participants in the Trial: 643.
Allocation: Randomized.
Primary Purpose of the Trial: Treatment of chronic constipation.
Inclusion Criteria for the Trial:
- Patient has completed the procedure of colonoscopy according to the criteria of the American Gastroenterological Association (AGA).
- Patient demonstrating continuous chronic constipation throughout the pretreatment phase.
- Patient has completed the protocol procedures with no significant clinical findings.
Exclusion Criteria for the Trial:
- Patient with a history of watery and loose stools.
- Patients either having symptoms or diagnosed with irritable bowel syndrome.
- Patients with any structural abnormality in the gastrointestinal tract or any disease or condition which can affect the gastrointestinal motility.
- Patient with any medical or surgical history of clinical significance which is not included in the protocol.
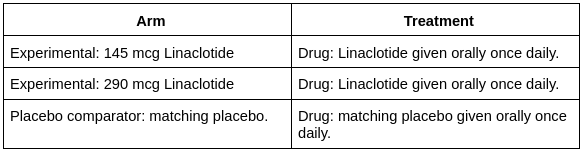
Primary Outcome of the Trial: Complete spontaneous bowel movement (CSBM) in a time frame from baseline to week 12. A week 12 CSBM was stated as a patient who had a CSBM weekly frequency rate greater than 3 and increased by one from the baseline for at least nine weeks out of 12 weeks. Complete spontaneous bowel movement (CSBM) is a spontaneous bowel movement that is supposed to be associated with the sense of complete evacuation.
-
A safety and efficacy trial of different doses of Linaclotide which is administered in children aged between 7 to 17 years of age and suffering from irritable bowel syndrome along with constipation.
Purpose of the Study: The study includes a screening period of up to four weeks and a pretreatment period of two to three weeks. Patients between the age of 7 to 11 years will be given oral liquid or solid oral capsules, while patients between the ages of 12 to 17 years will be given solid capsules orally. Children between the age of 7 to 11 years meet the entry criteria which is randomized to 1 to 3 doses of Linaclotide or placebo for around four weeks. Children ages 12 to 17 years will be randomized to 1 to 4 doses of Linaclotide or the placebo for about four weeks. This four-week study will assess Linaclotide's efficacy on the frequency of bowel movements and other symptoms of irritable bowel syndrome.
Number of Participants: 101.
Allocation: Randomized.
Primary Purpose: Treatment of irritable bowel syndrome.
Inclusion Criteria for the Trial:
- Patients should have a body weight of at least 18 kgs.
- Patients should meet the criteria of Rome III for children or adolescents with irritable bowel syndrome.
- Patients who meet the criteria of modified Rome III for children and adolescents with functional constipation.
- Patient who is ready to discontinue any laxatives which were used before the pretreatment visits.
- Patients who had an average of less than three bowel movements in a week during the 14 days before the randomization day.
Exclusion Criteria:
- Patients with more than one loose stool.
- History of any other condition that may lead to constipation may affect the trial's efficacy.
- Patients who required manual disimpaction anytime before the randomization.
- Patients who are not able to tolerate a placebo in the screening period.

Can Linaclotide Be Used for Weight Loss?
Linaclotide is not a weight loss drug and should not be used for this intention. Patients consuming Linaclotide may have weight loss due to side effects of diarrhea, but that does not mean the drug should be misused for weight loss.
Food Interactions of Linaclotide: Interaction of the drug Linaclotide with food can increase the drug's gastrointestinal side effects, leading to diarrhea, gas, and abdominal pain. So it is advised to take the drug at least half an hour before breakfast on an empty stomach.
Linaclotide and Pregnancy: The interaction of the drug with the fetus is still under study. However, animal studies reveal that the fetal effects of the drug are seen only in cases of maternal toxicity and with doses much more than the maximum suggested dose. Thus Linaclotide should not be used in pregnancy until the benefit of the drug outweighs the risk to the baby.
Linaclotide and Breastfeeding: The presence of drugs in breastmilk is still unknown, so it is suggested to discontinue the drug while breastfeeding.
Linaclotide in Geriatric Patients: The drug can be given to older patients suffering from irritable bowel syndrome. The doses should be administered according to the body weight and tolerance.
Difference Between Linaclotide and Laxatives:
Laxatives help to relieve constipation but in a different way than Linaclotide. Laxatives loosen the stool to prevent strain during bowel movements, whereas Linaclotide induces increased intestinal fluid secretion for the food movement. Therefore, laxatives are considered a short-term treatment for constipation. However, Linaclotide is a long-term treatment option for constipation and irritable bowel syndrome.













